
16 min
3
10.04.2022

Titration analysis is one of the essential types of quantitative analysis. Its main advantages are accuracy, speed of execution, and the possibility of using it to determine a wide variety of substances. The content substance's determination in the titration lab report was carried out due to the reaction precisely known as one substance with an unknown amount of another.
Typically, acid-base titration lab reports use solutions of substances. During titration, one substance's solution is gradually added to another substance's solution until the substances have completely reacted. The solution that poured in is called the titrant. The solution to which the titrant poured is called the titrated solution. The solution's volume to titrate that undergoes titration is called the aliquot part or aliquot volume. Check out our writing assignments guide to learn how to create an acid-base titration lab report.
How to Write an Acid-base Titration Lab Report
Acid-base titration lab report is one of the forms of material study that has found wide application in modern teaching methods. Depending on the number of participants in the experiment, classes divide into groups, units, and individuals. In the course of the research, it is necessary to follow the specified algorithm for performing experiments; otherwise, you may not register the passage of the process of interest or distort the result. The formal lab report submitted in printed form:
- typed in a word program, google docs;
- printed on sheets of A4 format, one side of the print;
- writing font Times New Roman;
- type size (font size) 14 points;
- line spacing 1.5 cm;
- from the left edge, the indent is 3 cm;
- from the right - 1 cm;
- top and bottom 2 cm each;
- page numbering begins with the title page, but the number is not put on the page itself and the content of the work;
- every application and graphic needs to be given a title.
What To Include In The Acid-base Titration Lab Report
The basic lab report consists of the following chapters:
- Title page.
- Description of the purpose of the work. When writing the work's goal, you need to indicate the laboratory's topic and list the main tasks assigned to the student.
- It is providing brief theoretical information. When listing brief theoretical information, you can give the necessary formulas, calculations. Here it is not necessary to indicate information that coincides with the textbook or methodology - it is enough to mention in general terms the basic concepts, laws, calculation tables, and formulas that the student used during the experiment. In terms of volume, this part should not exceed 1/3 of the entire document.
- Description of technical equipment and methods for experimenting. When describing the technical equipment and methods of experimenting, they talk about the progress of work, detail the experiment's course, the method of obtaining information, and the method of processing it.
- Results obtained during the experiment. In describing the results obtained during the experiment, you need to provide the most interesting data that the student received. Here we must not forget about specifying measurement errors.
- Analysis of the data obtained during the experiment. The section analyzes experimental data, detailed information indicated, and analyzes the results obtained and their interpretation based on the subject's laws being studied.
- Summarizing. In the summarizing section, conclusions should be drawn, supported by experimental action and theoretical knowledge.
How Long Should My Lab Report Be?
The student research project refers to small works. As a rule, it is no more than 10-20 pages:
What Is the Acid-base Titration Lab Report Purpose?
The chemistry lab report's primary goal is to learn how to prepare and standardize the acid-base titration method's titrant solutions. And also to master the technique of neutralization to determine the concentration of acids, bases, etc.
The neutralization method determines the concentration of acids, alkalis, salts that undergo hydrolysis, and substances that react with acids and alkalis. It is a fast and fairly accurate method for analyzing substances (both in aqueous and non-aqueous media).
This method based on the reaction of the formation of water molecules from hydronium and hydroxyl ions:
H3O + OH = 2H2O
In the neutralization method, solutions of strong acids (acidimetry) and alkalis (alkalimetry) use as titrants. Solutions of acids (usually hydrochloric or sulfuric) are prepared from concentrated solutions by the dilution method, and then their concentration is established using tetraborate or sodium carbonate.
Solutions of alkalis (sodium hydroxide, potassium hydroxide, barium hydroxide) are also prepared by diluting more concentrated solutions or by an approximate sample, then standardized against oxalic acid (or other standard acid solution).
Setting Up an Acid-Base Titration
An acid-base titration is based on the combination of H3O+ ions and hydroxide of OH- ions with the formation of low-dissociating water molecules. This method, using titrated solutions of alkalis, determines the amount or concentration of acids. With the help of acid solutions, the concentration of bases is found.
Since any external effect does not accompany the neutralization reaction, for example, a color change in the solution, the equivalence point is determined using indicators. Usually, indicators change color not strictly at the equivalence point, but with some deviation from it.
The titration endpoint does not always coincide with the equivalence point. It only more or less corresponds to the equivalence point. Therefore, even with the correct choice indicator, an error called the titration indicator error is allowed.
Alkalimetry is an analytical method by which the number of free acids or salts in alkaline compounds determine. To determine the amount of acid, a titrated solution of alkali NaOH or KOH use. Since an alkaline solution is unstable and often changes the titer, it is almost impossible to prepare an accurate titration solution by weighing all the components.
Alkalis are not components from which a titration base can be accurately prepared. Therefore, mainly acids are used as a basis for titration: oxalic or succinic. For each reaction, an indicator must be carefully selected, especially for salts formed during titration.
In the acidimetric titration of aqueous solutions, solutions of strong acids (HCl, less often HNO3 or H2SO4) use as titrants. However, the listed reagents do not have properties that would make it possible to prepare standard solutions only by an accurate sample. Thus, solid alkalis are hygroscopic and always contain carbonate impurities.
In HCl and other strong acids, the starting reagent is not a pure substance, but a solution with an inaccurately known concentration. Therefore, in the neutralization method, a solution is first prepared with an approximately known concentration, and then it is standardized.
Materials for a Titration Procedure
Equipment and reagents required for the titration lab report:
- On the student's table: pipettes for 5 ml and 10 ml (2 pcs in total), conical flasks (or cups) for titration of 50 ml or 100 ml (4 pcs in total), burette with a Mohr clamp (25 or 50 ml), a tripod with a clamp for fixing the burette, a rubber bulb (1 pc), a small funnel for filling the burette (1 pc), sheets of white paper (students bring 4 A4 sheets), phenolphthalein indicators, methyl red.
- On the teacher's table: fixed channels of acids, alkalis, and salts (for familiarization with students), a 1-liter volumetric flask, a funnel with a diameter of 60-80 mm, strikers for breaking the fixed channel, a titration kit (similar to a student's one).
- Task issuing table: solutions with different acid and alkali concentrations, titrated solutions of hydrochloric acid (0.1 M), and sodium hydroxide (0.1 M).
When carrying out titration, several general rules should be followed:
- All dishes used in the experiment should be thoroughly washed - the water should evenly wet the dishes' walls without leaving drops
- Volumetric dishes: (flasks, pipettes, burettes) must be pre-calibrated since the dishes' actual capacity often differs from the one indicated on the label.
- The glassware is weighed on an analytical balance for calibration, then filled with distilled water and weighed again.
- The mass of water determines the actual capacity of the dishes.
- Volumetric flasks and pipettes should not be held by the expanded parts, as the heat of the hands expands the glass, and the volume of the dishes may change.
- All solutions should have the same temperature of about 20°C.
Equivalence Point Indicators
A feature of audiometry and alkalimetry is a change in the solution's pH during the titration. The medium can be neutral, slightly acidic, or slightly alkaline at the point of equivalence.
In this regard, three main causes can be distinguished:
1.Titration of a strong acid with a strong base:
HCl + NaOH → NaOH + H2O
The product of this reaction is a salt that does not undergo hydrolysis. This means that the equivalence point, in this case, will be in a neutral environment at pH = 7.
2.Titration of a weak acid with a strong base:
CH3COOH + NaOH ↔CH3COONa + H2O
The resulting salt in an aqueous solution will undergo hydrolysis by the anion:
CH3COOH + NaOH ↔CH3COONa + H2O
As a result, even after the initial substances' complete consumption, the medium's reaction at the point of equivalence will be weakly alkaline (pH7).
3.Titration of a weak base with a strong acid:
NH3H2O + HCl ↔NH4Cl + H2O
The resulting salt in an aqueous solution will undergo cation hydrolysis: NH4 + HOH↔NH3H2O + H
Interactions between acids and bases are usually not accompanied by any external effects. Most acid and alkalis solutions are colorless, as are the solutions formed by the interaction of salts.
The titration's endpoint is detected potentiometrically, conductometric, spectrophotometric, or other instrumental methods and visually.
Acid-base indicators are widely used for visual fixation of the equivalence point. The most important acid-base indicators:
Estimating the Equivalence Point
The parameter characterizing the acidic, basic (alkaline) or neutral nature of an aqueous solution is called pH (pH):
pH 7: acidic solution
pH = 7: neutral solution
pH 7: basic (alkaline) solution
Determination of pH directly gives information about the presence of certain ions in the solution: hydrogen ion H+ and hydroxide ion HO-:
pH 7: The solution is acidic and contains more hydrogen ions H+ than hydroxide ions HO-.
pH = 7: Neutral solution containing the same amount of hydrogen ions H+ as hydroxide ions HO-.
pH 7: Basic (alkaline) solution with more hydroxide ions HO- than hydrogen ions H+.
The pH is related to the determination of the concentration of H+ ions. These ions, like hydroxide ions, react with many substances, including water. Strong acids or strong bases (alkalis) are particularly corrosive. They must be handled with great care. The pH value: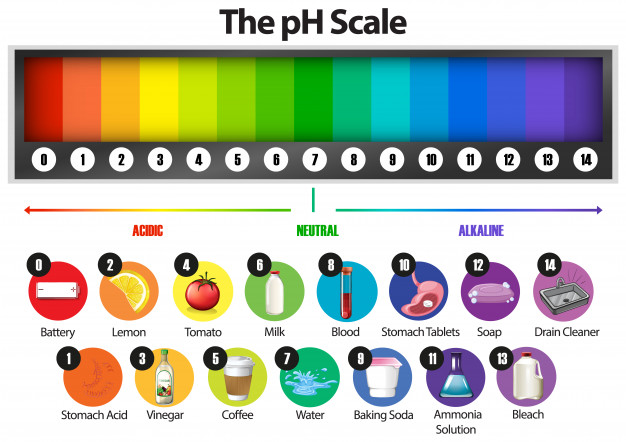
Titration Procedure
Experience 1. Acid-base titration. Determination of the concentration of alkali solution.
The alkali solution's concentration is determined by direct titration with a standard acid solution in the presence of methyl red or methyl orange indicator.
We are completing the work. Get an alkali solution of unknown concentration. In a conical titration flask, place an aliquot of the dispensed alkali solution (5-10 ml). Add 4-5 drops of methyl red indicator. Fill a clean burette with a standard acid solution and titrate the alkali solution until the yellow color changes to orange-yellow. Titrate 3-4 samples, find the average volume of the titrant solution. The results are recorded in the laboratory journal. For a more accurate determination of the end of the titration:
- place a sheet of white paper under the titration flask;
- prepare a witness solution.
Calculation of results. An alkali solution's molar concentration is calculated because solutions of the same molar concentration react in volumes proportional to the stoichiometric coefficients in the reaction equation.
Work report. Based on the information provided in the STATISTICAL PROCESSING OF RESULTS section, perform statistical processing of the determination results. All calculations should be performed transparently, indicating the initial calculation formulas, then formulas with the variables' substituted numerical values, and write down the answer indicating the dimension. All intermediate calculations should be performed with an accuracy of two to three orders of magnitude higher than the original presented data.
Experience 2. Acid-base titration. Determination of the concentration of the acid solution.
The acid solution concentration can be determined by direct titration with a standard alkali solution in the presence of the phenolphthalein indicator. We are completing the work. Get an acid solution of unknown concentration. Place an aliquot of the dispensed acid solution (5-10 ml) into a conical titration flask. Add 4-5 drops of phenolphthalein indicator. Fill a clean burette with a standard alkali solution and titrate the acid solution until a stable pink color appears. Titrate 3-4 samples and find the titrant solution's average volume in the laboratory journal's results record. For a more accurate determination of the end of the titration:
- place a sheet of white paper under the titration flask;
- prepare a witness solution.
Calculation of results. The acid solution's molar concentration is calculated based on the basis that the same molar concentration solutions react in volumes proportional to the stoichiometric coefficients in the reaction equation.
Work report. Based on the information presented in the STATISTICAL PROCESSING OF RESULTS section, perform statistical processing of the determination results for both experiments. All calculations should perform transparently, indicating the original calculation formulas, then formulas with substituted numerical values of variables, and the answer indicating the dimension. All intermediate calculations should be performed with an accuracy of two to three orders of magnitude higher than the original presented data.
What Methods Should You Use?
In the lab reports, several titration methods are distinguished:
- Indirect titration methods, the analyte solution is directly topped up with the titrant from the burette.
- Back titration methods (residual method) use two standard solutions - primary and auxiliary. A significant excess of the basic standard solution is added to the analyzed solution, and then its excess is titrated with a standard auxiliary solution.
- Titration of a substituent or substituted by titration (indirect titration) when a special reagent is added to the analyte, which quantitatively interacts with it. Then, one of the reaction products titrates with a standard solution.
- In some cases, a reverse titration use, in which a portion of a standard solution takes with a pipette, titrates with an analytical solution. When a fragile substance is analyzed in the air (oxidizes, absorbs CO2, H2O, etc.), the method is used.
Advantages of the Acid-base Titration Method
All definitions based on the reaction belong to the acid-base titration method: H + OH = H2O
Using a titrated solution of some acid, this method can carry out a quantitative determination of alkalis or, using a titrated alkali solution, quantitatively determine acids.
Using this method, several other determinations are carried out, such as determining some salts. Like Na2CO3 and Na2B4O7, they have a strongly alkaline reaction due to hydrolysis. Therefore, they titrate with acids, determination of water hardness, determination of ammonium salts, determination of nitrogen in organic compounds, etc. This method considers the best for the lab report because it is the easiest among the others mentioned above.
The main working solutions in this method are the acid solution (usually HCl or H2SO4) and alkali solution (usually NaOH or KOH). These substances do not meet the requirements for standard substances. Therefore, their titrated solutions cannot be prepared based on an exact sample of the substance, the solution of which dilutes to a certain volume. The concentration of such solutions has to be determined by titration.
What Are the Other Methods of Chemical Analysis?
Chemical analysis methods can be classified based on different principles or approaches. They can be classified according to the phenomenon used to obtain analytical information about the composition of substances. According to this classification, methods divide into four groups:
- Chemical methods using chemical reactions.
- Physicochemical methods based on the use of the interaction of a substance with physical fields (electromagnetic, electric, etc.), this action is preceded by a chemical reaction.
- Analysis methods use the same interactions without a chemical reaction.
- Biological methods are based on biochemical processes and interactions with microorganisms.
Thus, chemistry applies a large arsenal of methods related to other sciences to solve its problems. This is a feature of modern chemistry - the versatility of methods for solving its problems, the above methods are suitable for a science lab report on the subject.
Where Is an Acid-base Titration Technique Used In Production?
Acid-base titration methods use in production control in the chemical, textile, food industries, in the technology of plastics and fibers, fertilizers, in metallurgy, etc. The method under consideration is important for sanitary and hygienic environmental objects, since industrial effluents may contain acidic or alkaline waste. This method is also widely used in the technological control of winemaking to determine titratable and volatile acids - tartaric, malic, succinic, citric, and carbonic acids.
Insanitary and hygienic practice, acid-base titration is used to determine the carbonate hardness of water to assess food quality: milk, cottage cheese, bakery products, in which organic acids are formed during storage as a result of spoilage.
In the pharmaceutical industry, pharmacies use to determine the concentration in several drugs of an acidic or essential nature.
In biochemical studies, clinical practice, using acid-base titration, the buffer capacity of biological fluids, the alkaline reserve of blood and plasma, and gastric juice's acidity determine. In the latter case, its active and general acidity is distinguished.
Don’t Forget About Revision
Every research in the laboratory is mini-scientific work. Accordingly, the preparation must be serious. Certain difficulties in solving laboratory work arise at the stage of information search and analysis. Many skip this stage and start working on the practical part. This leads to the fact that students do not understand the experiments' essence and the result.
Difficulties also appear when performing the practical part because the student does not meet all the experimental conditions. For example, it was necessary to heat the substance to 50 degrees, but it turned out to 47. This is how false results appear that differ from the expected ones. Therefore, check the documents before submitting; this can be done using special programs.
To edit a document in Microsoft Word and Google Docs, the Edit / Find and Replace command provide. The provided ability to find and replace text speeds up editing (editing) existing documents. It can be useful when finding and replacing certain formatting parameters (such as APA style for a lab report, MLA, ASA, etc.), special characters (for example, tabs or optional hyphens), and other document objects.
Things to Avoid
- Carefully indicate the definition of basic concepts, laws, rules, fundamental provisions of the theory, formulas, generally accepted symbols for the designation of physical quantities, and measurement units. Otherwise, you will be downgraded for making a mistake in such an important matter.
- Highlight the main thing in the answer, do not use information that does not relate to laboratory work.
- Apply all your knowledge to solve problems and explain physical phenomena, or incorrectly formulated questions of a problem or incorrect explanations of its solution's course will lead to the fact that all the work is done in vain.
- It is better to build graphs and schematic diagrams than to write the data in words.
- Use examples of labs to avoid mistakes, but do not copy. Your work must be unique without plagiarism.
- Careless handling of laboratory equipment or measuring instruments can lead to your being excluded from laboratory work.
- Do not violate safety requirements, discipline when performing a task, experiment.



